
What is a Colloidal Solution? In a colloidal solution, the particles of a colloid are uniformly spread throughout the solution. The table below summarizes the properties and distinctions between solutions, colloids, and suspensions. Because the dispersed particles of a colloid are not as large as those of a suspension, they do not settle out upon standing. The particles are spread evenly throughout the dispersion medium, which can be a solid, liquid, or gas.  
What is the difference between a suspension and a colloid? What are examples of a suspension?Įxamples of a suspension include the following: Whole milk, peanut butter, some salad dressings, hot chocolate, oil paint, muddy river water. 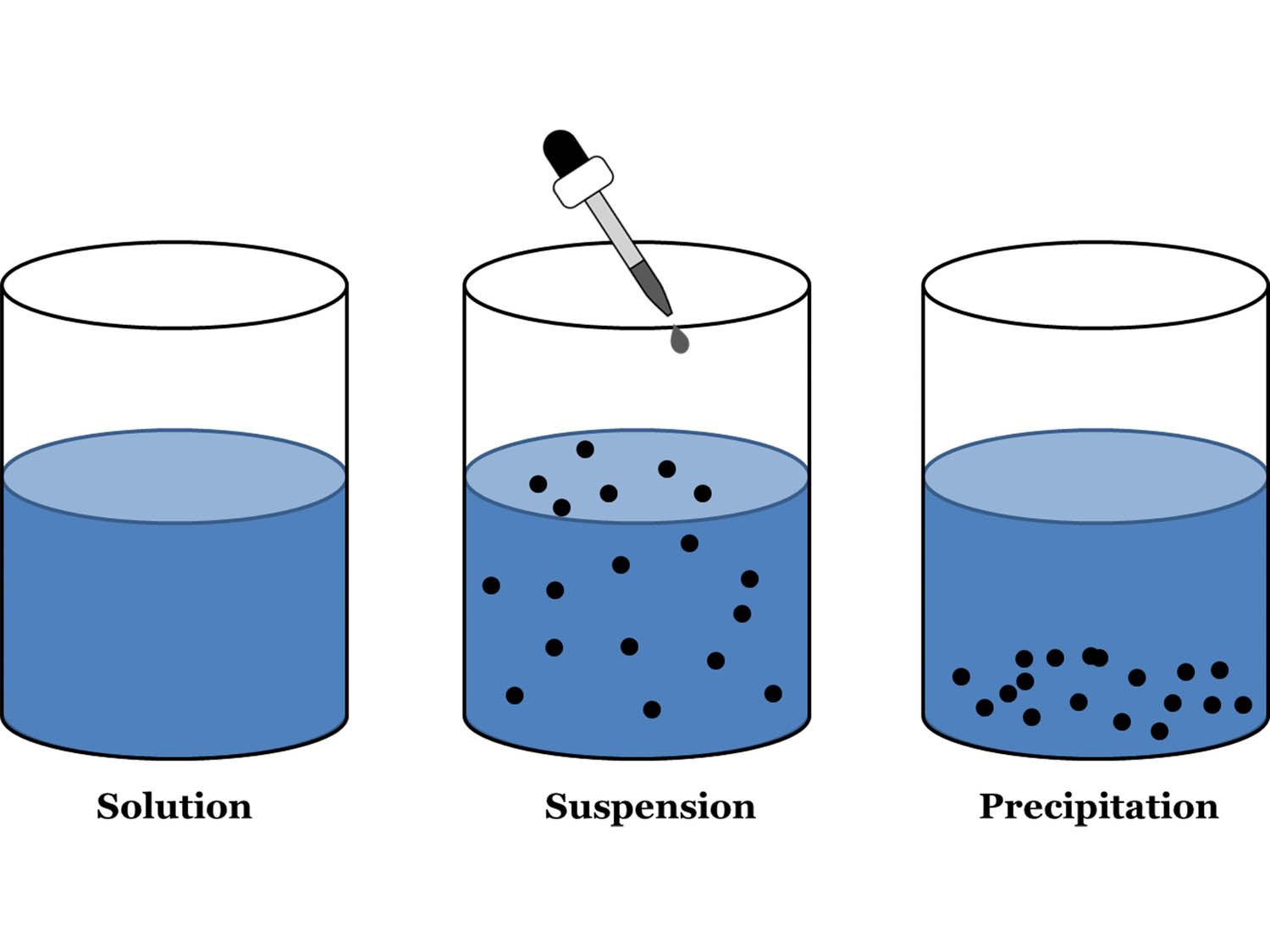
Milk is a colloid, with tiny globs of butterfat suspended throughout the liquid. Is syrup a colloid suspension or solution? When you add oil to vinegar and shake hard, you’ll see small pockets of oil in the vinegar and vice versa, all mixed together in a suspension. Vinegar is a solution of acetic acid dissolved in water – notice that you can’t see any particles in the liquid. Is vinegar solution colloid or suspension? If you mix oil and water and shake them a cloudy suspension is formed. 6 What is the difference between a suspension and a solution?Įmulsions are a type of colloid An example would be oil and water.2 Is vinegar solution colloid or suspension?.1 Is oil a colloid suspension or solution?.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
